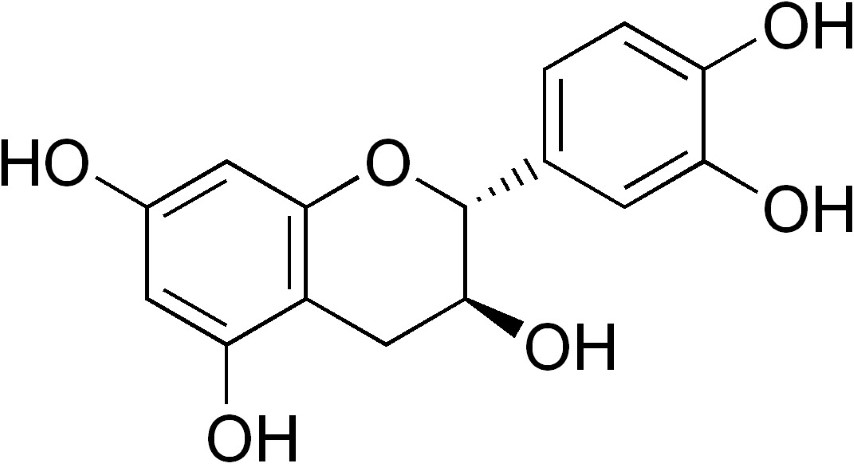
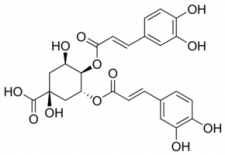
Chlorogenic Acid in Coffee: Chlorogenic acid (CGA) is a naturally occurring compound found in coffee beans, and it plays a significant role in determining the flavor profile and potential health benefits of coffee. In this article, we will discuss the presence of chlorogenic acid in coffee and its impact on taste, roasting, and health.
Overview of Chlorogenic Acid in Coffee
- Impact on Taste: Chlorogenic acid contributes to coffee’s acidity, which is an essential factor in the overall taste profile. Acidity adds brightness and complexity to the coffee, balancing out the other flavor components such as sweetness, bitterness, and body. Generally, coffee beans with higher levels of chlorogenic acid tend to have a more pronounced acidity, resulting in a vibrant and lively taste.
- Roasting and Chlorogenic Acid: The roasting process directly impacts the levels of chlorogenic acid in coffee beans. As the beans are roasted, the chlorogenic acid content decreases, reducing the perceived acidity in the brewed coffee. Light roasts generally have higher levels of chlorogenic acid, resulting in a brighter and more acidic flavor, while darker roasts have lower levels, leading to a bolder, less acidic taste.
- Potential Health Benefits: Chlorogenic acid has been studied for its potential health benefits, including antioxidant, anti-inflammatory, and antimicrobial properties. Research suggests that it may help in regulating blood sugar levels, reducing blood pressure, and aiding weight management. However, it’s essential to note that the overall health impact of coffee consumption depends on various factors, including the amount consumed, individual tolerance, and preparation methods.
In conclusion, chlorogenic acid plays a crucial role in shaping the taste of coffee and offers potential health benefits. Understanding its presence in coffee beans and how it’s affected by roasting can help coffee enthusiasts make informed choices to suit their preferences and health needs.
Chlorogenic Acid in coffee
Qualitatively, quinic and caffeic acids have been related to the higher degrees of astringency, bitterness, and body in darker roasting methods.
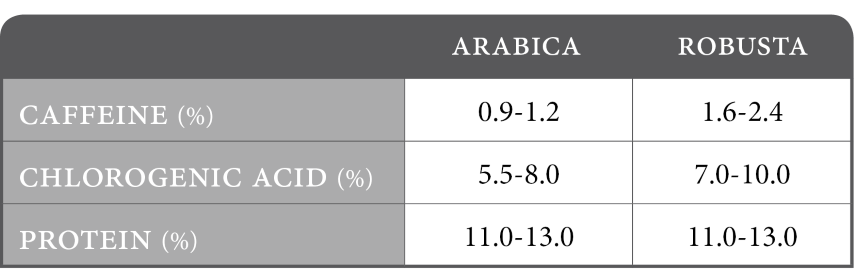
Selected composition for raw coffee (%):
In Arabica and Robusta, coffee contains the highest percentage of chlorogenic Acid of any species in the plant kingdom, at 6-10 percent dry weight.
Changes in climatic conditions, stress on the plant, and insect infestation are among the causes that cause CGA formation. Robusta, cultivated under more challenging circumstances, has approximately twice as much CGA than arabica.
Also, the synthesis of CGA and caffeine are tightly related, such that when CGA concentrations grow, so do caffeine concentrations.
In actuality, there are over a dozen isomers of chlorogenic Acid, each with different sensory properties. CGA-3 predominates in coffee with different amounts of mono, di, and feruloyl quinic acids.
According to recent research, the “di-CGA” version of the Acid may be responsible for the bitter/metallic flavor notes seen in certain coffees. In the case of robusta, which has more significant amounts of di-CGA and a harsher taste profile, this may very well be the case.
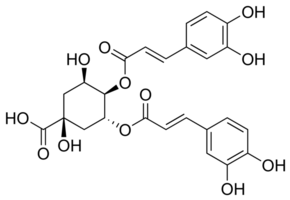 |
| 3,4-dicaffeoylquinic acid (di-CQA) |
There are 200 to 550mg per cup (6oz) of coffee, significantly surpassing the antioxidants found in green tea. As a result, coffee tends to be richer in simple phenolics than tea, whereas tea tends to have more catechins.
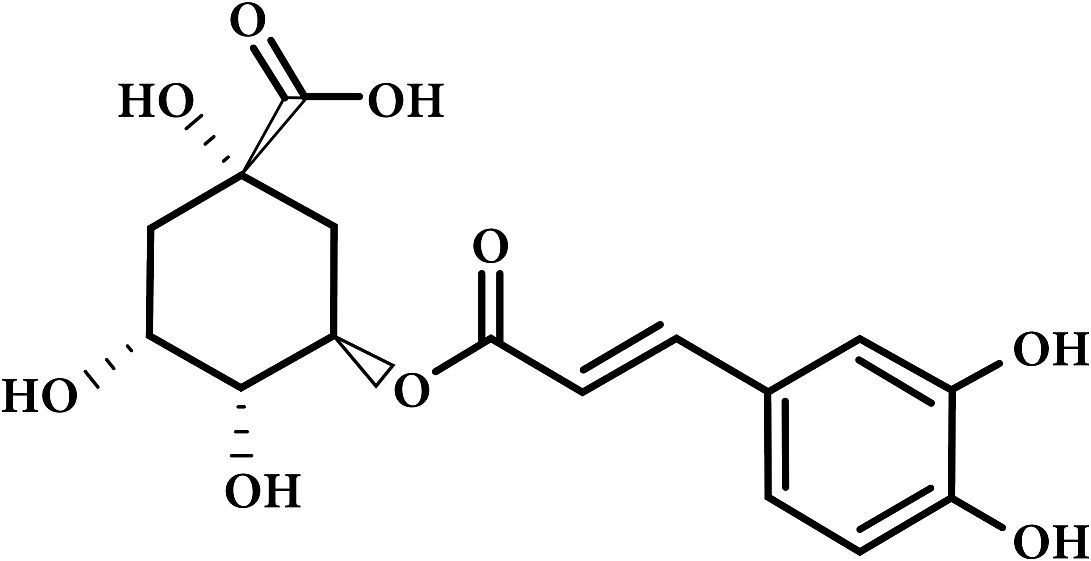
Chlorogenic Acid