
Chemical Changes During Coffee Roasting: Hundreds of chemical reactions occur when the coffee beans go through the roaster’s heat, with some molecules breaking down, others denatured into other compounds, and countless new combinations produced.
This, plus a series of physical changes, will form in the coffee bean’s unique flavors. So let’s learn about Chemical changes during coffee roasting with Helena.
Every reaction involving coffee in a roaster is called pyrolysis in chemistry. When organic material reaches the decomposition temperature, it undergoes reactions that create volatile chemicals and leave a carbon residue.
We established a limit during the roasting procedure to avoid charring the beans. But enough for them to undergo pyrolysis-induced chemical reactions and create taste molecules. Here are the fundamental chemical processes that go into making your morning cup of coffee:
The response of Maillard
Maillard’s chemical process creates roasted coffee’s characteristic flavor and dark color. The responses are named after the French physician Louis Camille Maillard, who initially characterized them in 1910.
When temperatures surpass a threshold in a roasting oven, the process begins at around 140°C to 160°C. Caramelization processes begin at 170°C, burning off any leftover sugar.

There are several methods for the Maillard process in coffee beans when amino acids and sugars are present, and we will end up with n two distinct taste compounds. Roast tastes, toast, and other similar goods are the most well-known.
The Maillard reaction is unique in that all its products may react with other free amino acids to produce other free amino acids. Melanoidin is a dark brown substance with a roasted, malty, bitter, burned flavor. It helps to develop and stabilize the crema in Espresso, which gives your coffee a rich taste.
The process of caramelization
Unlike the Maillard process, caramelization is pyrolysis at around 170°C (338°F). Due to the high temperature, long, complex carbohydrate chains will break down into hundreds of new, smaller molecules, adding a substantial degree of bitterness, sourness, and fragrance.
This reaction will take place continuously until the end. The roasting process also contributes to the sweet aromas in coffee, such as caramel, toast, almonds, etc.
It’s also worth noting that the amount of sucrose sugar in a coffee impacts the sweetness and richness of the coffee after roasting and the acidity because sucrose caramelization produces acetic acid.
As a result, we constantly urge coffee producers to harvest at the right stage of maturity, with the maximum possible sugar content, so that the Maillard & Caramel reaction team has plenty of raw materials.
Acidic decomposition
Alternatively, the acidity gives coffee vitality, refinement, complexity, and brightness. Even though many coffee users believe acid makes coffee bitter or unpleasant, coffee without acid is “empty” and dull.
Cold-brew coffee can be enjoyed to reduce the amount of acid in the cup. However, this effectively removes the laborious balancing acidity throughout the roasting process.
When it comes to acidity, we must remark that chlorogenic acid is by far the most frequent acid in raw coffee beans, with a CGA concentration of 6% to 8%, which is greater than any other plant.
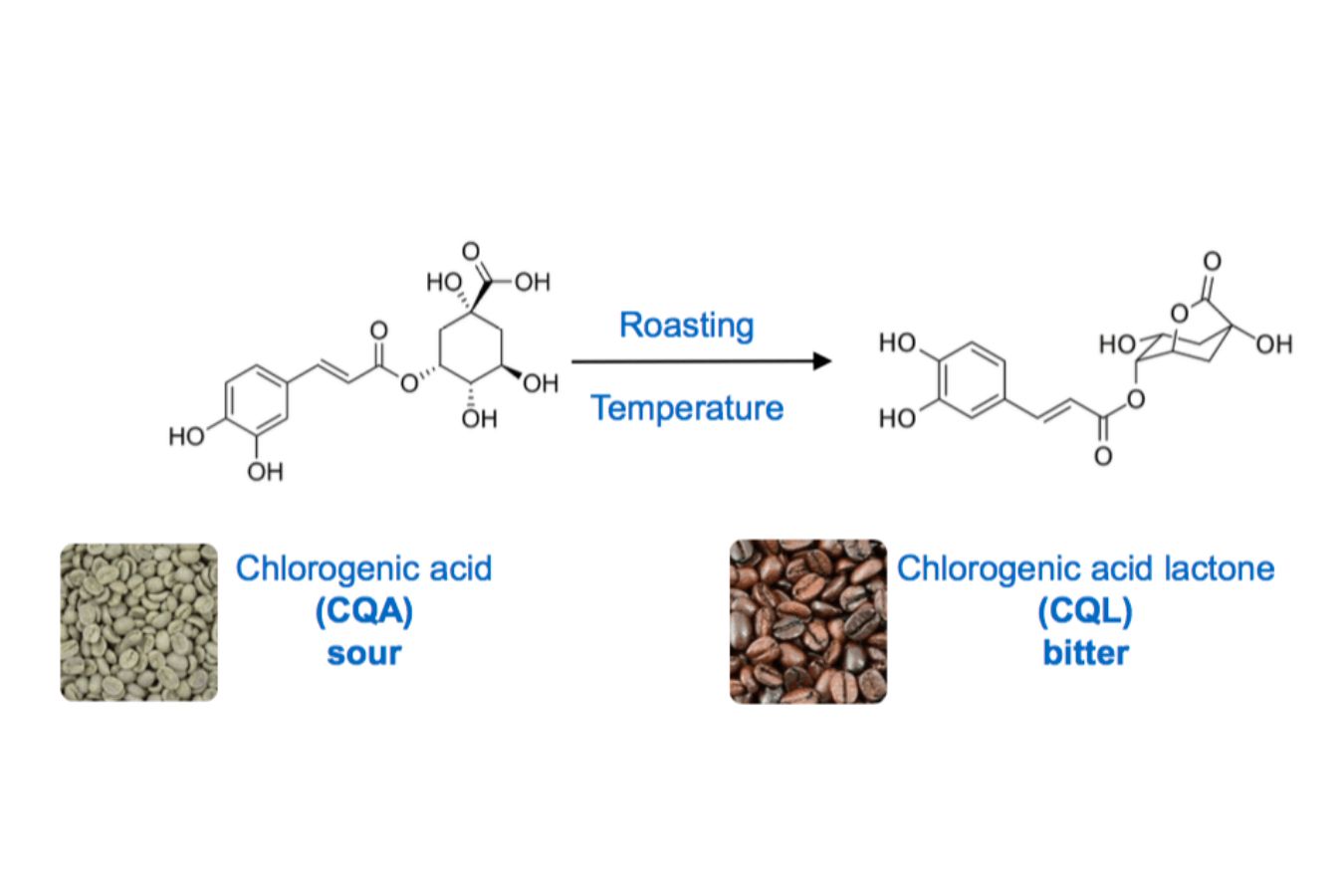
The roasting process continually breaks down CGA depending on temperature and duration; in light roasts, 50% of CGA is lost, whereas in dark roasts, only 20% is lost. CGAs do not evaporate; instead, they degrade into quinic and acids, two phenolic chemicals that add to the bitterness and body of coffee (Scott Rao, 2014).
Flavor compounds are formed.
After roasting, about a third of the mass of coffee is water-soluble. Meanwhile, the ideal extraction rate is around 19% – 22%, equivalent to roughly 55% – 60% of total soluble matter in coffee plus a minor quantity of lipids and broken cellulose pieces. All of these solutes and the pleasant scent result from one of the following transformations.
Volatile compounds
When the moisture content of the coffee falls below 5%, a quick surge in volatile aromatic compounds occurs, producing the ” coffee fragrance ” a few minutes after the roasting process begins.
The production of aromatic molecules is aided by the Caramelization and Maillard processes and the breakdown and metabolism of amino acids, sugars, phenolic acids, and lipids.

- The aroma of green grapes is aldehydes.
- Furans are a component of the caramel taste.
- Pyrazines are a kind of pyrazine with an earthy aroma.
- The scent of guaiacol is strong and smoky.
- Some sulfur compounds, such as 2-furfurylthiol, provide the distinctive scent we refer to as “coffee roasting,” but others do not.
It’s worth noting that the scent level will be highest for light to medium roasts. As the meat roast darker, the heat eliminates the smells it produces, leaving a smoky and spicy odor behind. At the same time, as the cellulose structures become weaker and more porous as the roast gets darker, the capacity to retain scent will deteriorate.
Compounds that aren’t volatile
Chemicals that are stable at room temperature are referred to as “stable at room temperature.” While some of these compounds are produced and change throughout the roasting process, others do not. The majority of non-volatile chemicals contribute to coffee’s taste (flavor).

Caffeine, which is responsible for some bitterness, sugar for sweetness, and lipids for bodily sensations are examples of non-substances. Acids and melanoidin chemicals generated in the Maillard reaction are examples of non-substances.
When it comes to caffeine, contrary to widespread assumption, deeper roasting lowers the bulk of the beans owing to dryness, not the caffeine concentration (since caffeine is more stable at heat than roasting).
As a result, the darker the roast, the greater the caffeine/weight ratio; hence the darker the roast, the higher the caffeine content for the same volume of coffee.
Reference source:
- The Coffee Roaster’s Companion (Scott Rao, 2014)
- www.perfectdailygrind.com What Happens During Coffee Roasting: The Chemical Changes
- www.baristahustle.com/ What is the Maillard Reaction and Why is it Important?


