
The Crema Stabilization Chemistry – So yet, no comprehensive research on the chemical ingredients responsible for the creation and stabilization of espresso crema has been published.
Nunes et al. (1997) discovered that foamability increased linearly with a roasting degree. They also found a link between the amount of protein in the infusion and the outcome.
The amount of the polysaccharides galactomannan and arabinogalactan in espresso coffee was found to be related to foam stability. Total solids, pH, lipids, protein, and carbohydrate content were among the other dependent variables found.
The roasting procedure created complexes between polysaccharides, proteins, and phenolic chemicals, which were found to have a strong association between foam stability and high molecular weight molecules (Nunes and Coimbra, 1998).
Recent activity-guided research on foam-active components in green and roasted coffee has revealed new information about the molecular structures of the complex compounds that cause crema creation.
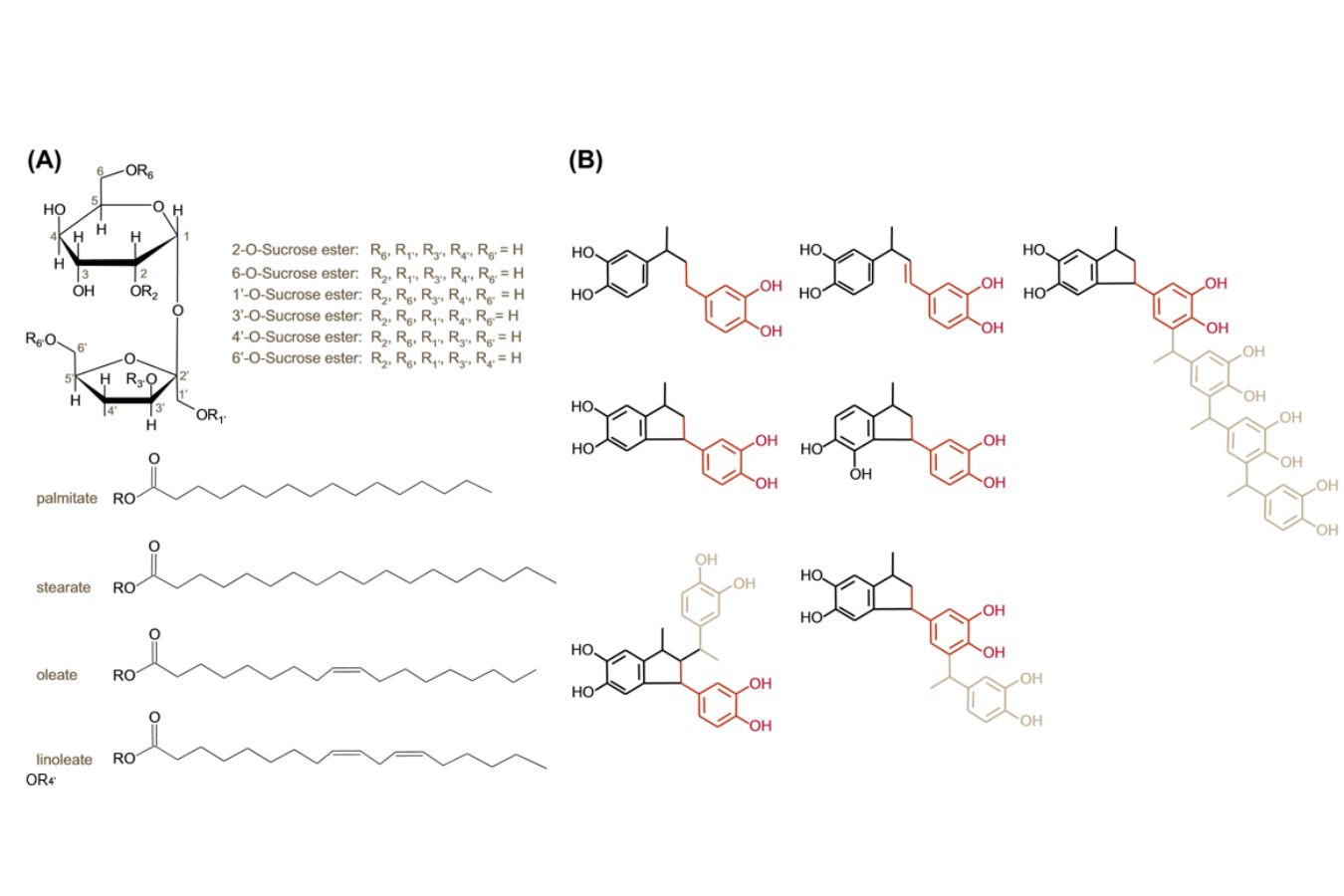
Although sucrose fatty acid esters (Fig. 17.1A) were shown to be the primary foam-active agents in green coffee, low- and high-molecular-weight 4-vinyl catechol oligomers (Fig. 17.1B) were discovered to be the essential foam-active components in roasted coffee’s exquisite crema (Unpublished data Kornas and Hofmann et al.)
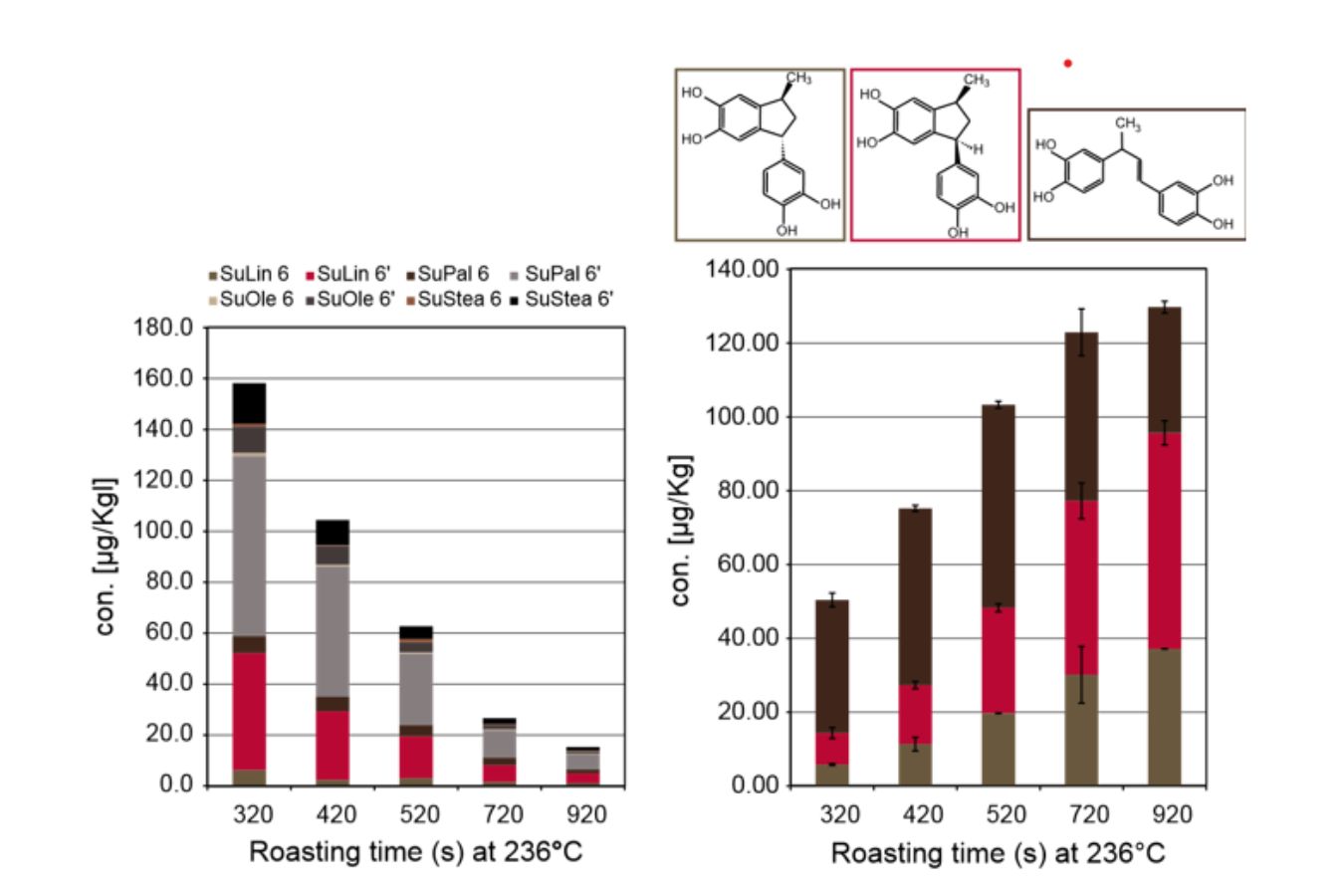
The sucrose esters were shown to be natural compounds in green coffee. They gradually deteriorated with increasing roasting time, according to a quantitative study of these foam modulators using liquid chromatography-tandem mass spectrometry (MS/MS) (Fig. 17.2A). The 4vinylcatechol oligomers, on the other hand, were shown to be formed as the roasting duration increased (Fig. 17.2B).
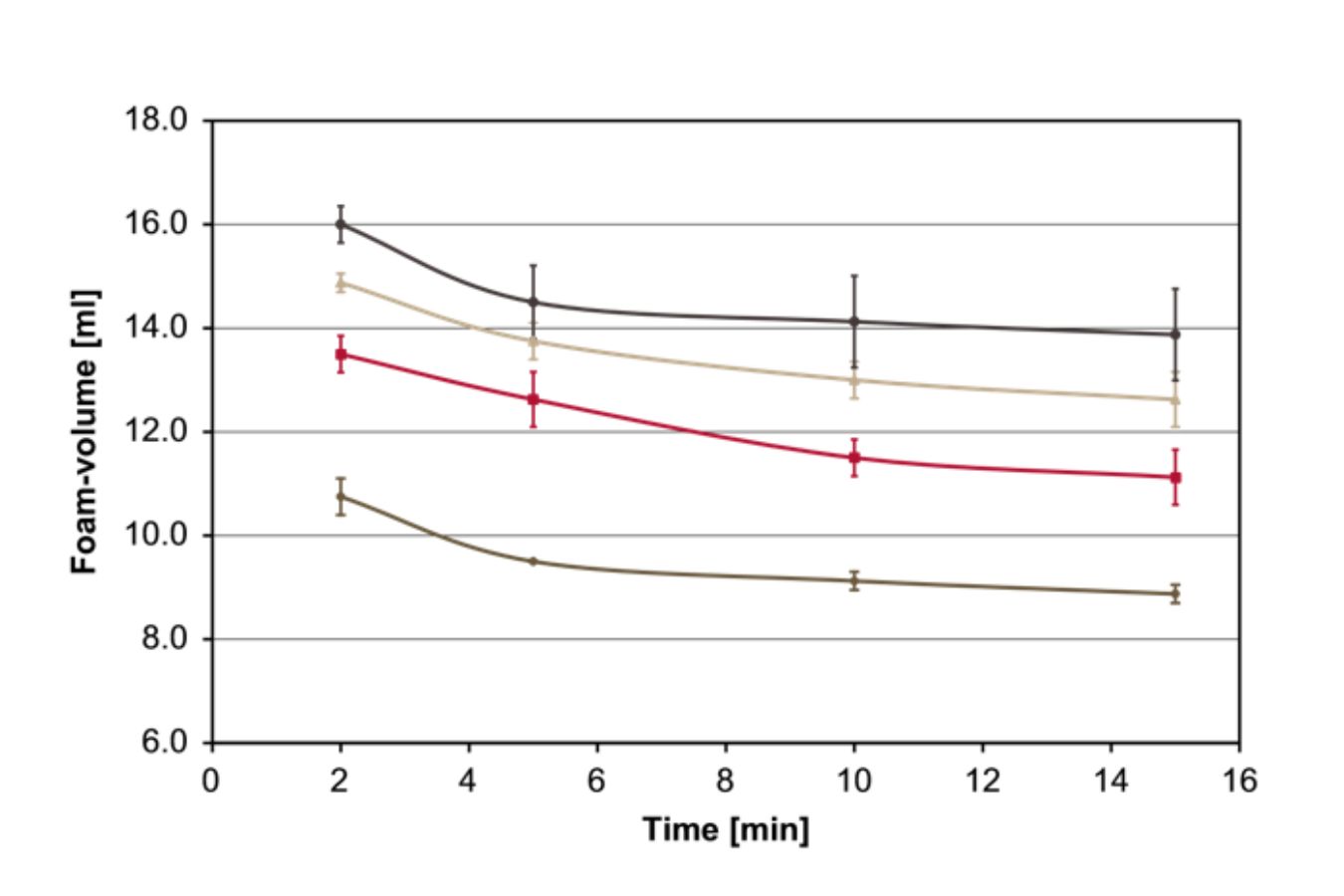
Caffeic acid was thermally treated for 20 minutes at 220C and then added to espresso coffee beverages in concentrations of 0.007%, 0.035 percent, and 0.07 percent before ultraturrax-mediated crema generation, as low- and high-molecular-weight 4-vinyl catechol oligomers were found to be generated by thermal degradation of chlorogenic acids and caffeic acid, respectively.
With higher degrees of roasted caffeic acid, the foam volume increased significantly; for example, when the coffee beverage was spiked with 0.07 percent burned caffeic acid (Fig. 17.3), the foam volume increased more than 60%.
These findings showed for the first time that low- and high-molecular-weight 4vinylcatechol oligomers, formed when caffeic acid moieties are roasted, are essential contributors to the crema of roasted coffee beverages.
To determine the effect of the foam’s active components on the foam structure of the crema, the foam was collected from espresso coffee spiked with sucrose palmitate (0.05%) or roasted caffeic acid (0.007%), as well as nonspiked espresso (control), and evaluated using a foam scan apparatus with a camera.
The foam images in Fig. 17.4 did not show any variations in the early stages of roasting. However, there were significant differences in the foam structure after 300 seconds.

The foam structure of coffee and coffee spiked with roasted caffeic acid was comparable, with smaller bubbles and more liquid between the bubbles. However, the foam formed with coffee spiked with sucrose-palmitate has a giant bubble with less liquid phase between the dots.
In contrast to the foam of coffee spiked with fatty acid sucrose esters, which is coarser and dryer, the foam of coffee spiked with roasted caffeic acid was comparable to the original espresso coffee foam.
According to our findings, the 4-vinyl catechol oligomers, not the sucrose esters, definitely play a crucial role in producing the fine-structured crema of quality espresso.
These findings suggest that because Robusta coffee is higher in caffeic acid and chlorogenic acid than Arabica coffee, it will promote the formation of high- and low-molecular-weight 4-vinyl catechol-based surfactants during roasting, which, in combination with the role of pressure and CO2, could explain the foam-promoting role of Robusta coffee in espresso blends.