Ethyl Acetate (EA) – Decaffeinated Solvent: Ethyl Acetate (EA)- A naturally present Ester (found in fruits and vegetables) can be isolated and used as a solvent to bind and remove caffeine from coffee beans (see decaffeination process). Coffee terms about Processing | The approach you should know are as follows:
- Fermentation
- Fresh Coffee
- Methylene chloride (MC) – Decaffeinated Solvent
- Natural process – Dry processing
- Raw processed – Natural processing
- Semi Washed – Semi-Wet Processing
- Sorting / Grading – Particle Classification
- Vacuum Cham – Vacuum Chamber
- Washed Coffee – Wet Processing
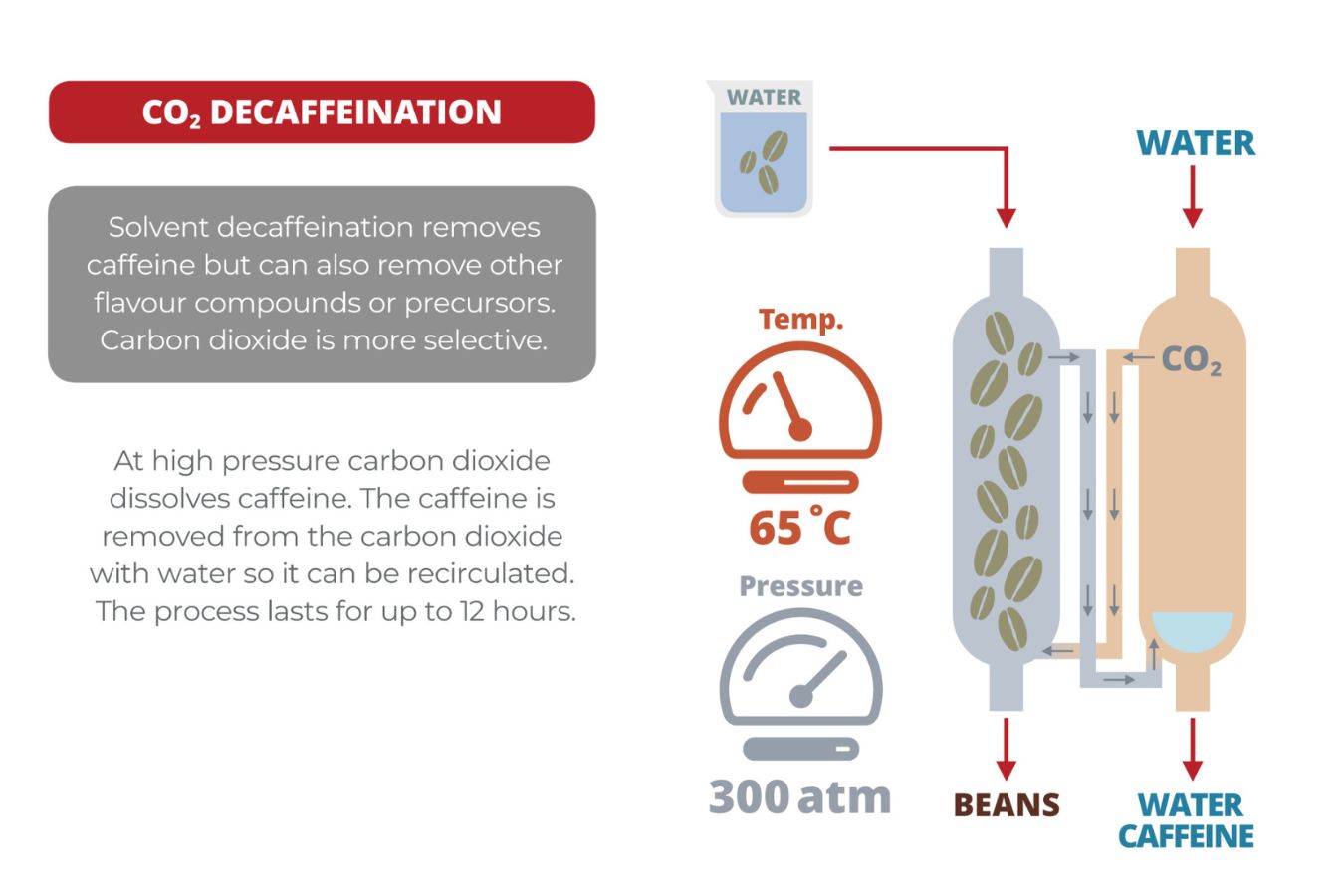
Ethyl Acetate – EA Decaffeination Process
First, the coffee is classified and steamed for 30 minutes under low pressure to open the porous holes of the coffee beans and prepare for the decaffeination process. Coffee is placed in the solvent of the water mixed with ethylene acetate, at which point EA will begin to bind to the salt of chlorogenic acid inside the seed.
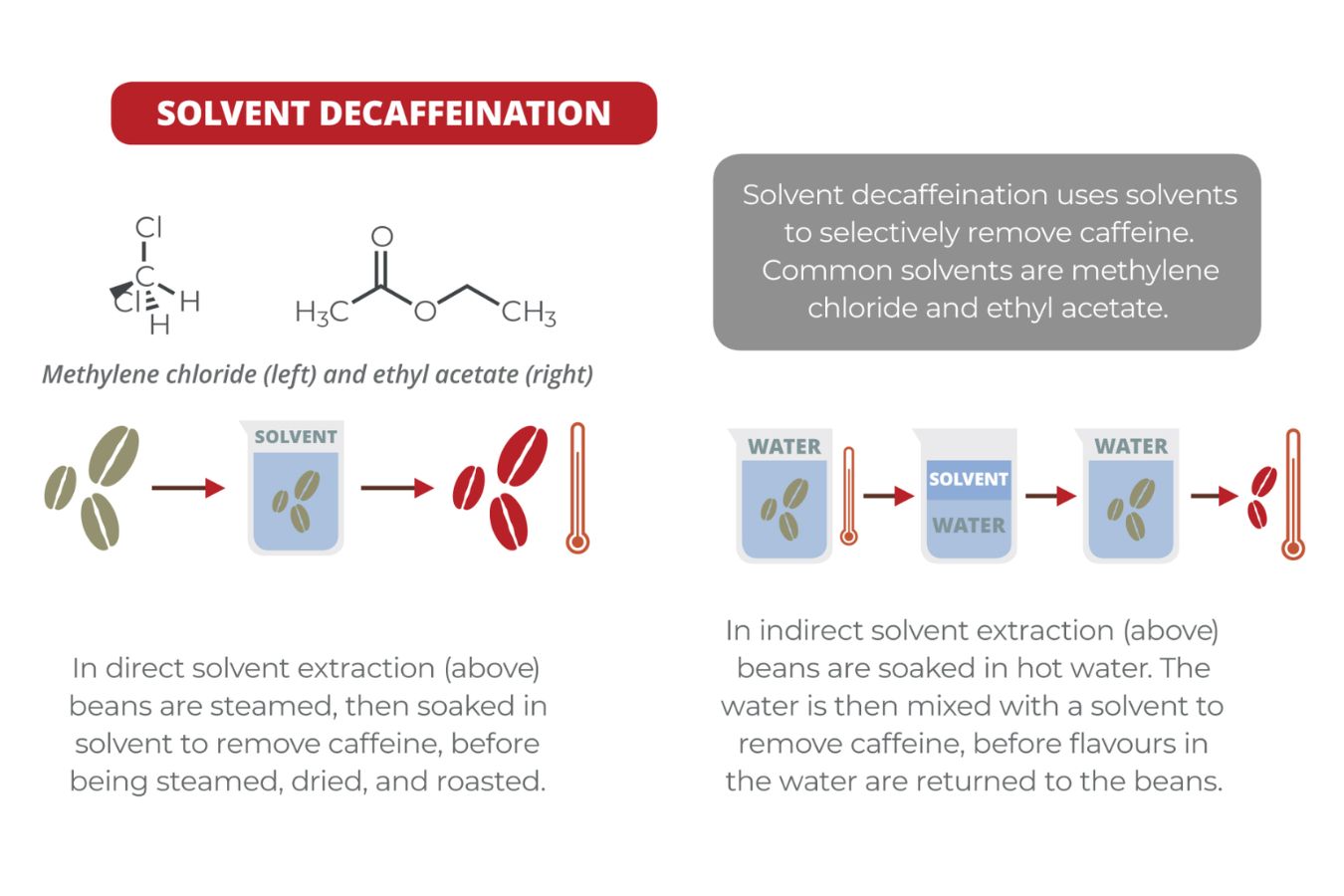
In the roots, caffeine is present in the form of salts chlorogenic acid. The tank will be discharged and re-performed within eight hours until caffeine is no longer detected.
The seeds are steamed again to remove traces of ethyl acetate, although EA is only harmful to humans in very high amounts (400 parts per million or more). The coffee is then dried and polished, ready for consumption.